Part 1: Pharmaceuticals
The US EPA released its draft contaminants candidates list (CCL6) on 02-April-2026. It contains a long list of individual contaminants including industrial chemicals, flame retardants, pesticides, ubiquitous emerging contaminants such as Bisphenol A and many more that were also included in the CCL5. While this alone would be several interesting projects on occurrence fate and transport, it is always the chemical groups that attract the most attention. In CCL6, 4 groups were included:
PFAS: The list excludes PFAS already subject to regulation (PFOS and PFOA essentially) and continues on from CCL5, which introduced to the US the concept of looking at PFAS as a class
Microplastics: Identified as a significant data gap, and there are other funding announcements on microplastics, watch this space.

Disinfection byproducts: 27 unregulated brominated haloacetic acids, haloacetonitriles, halonitromethanes, iodinated trihalomethanes, nitrosamines, and others. Expanded DBPs have been on our radar for a long time, let’s se
Pharmaceuticals: Substances that meet the definition for a “drug” under the Federal Food, Drug, and Cosmetic Act (21 U.S.C 321)
We have discussed microplastics extensively recently and, especially in drinking water, there are good options unless we have to go lower than 10um, when it starts getting progressively more difficult and expensive. DBPs are a topic of whole other discussion.
Let’s talk pharmaceuticals first, part of that large umbrella of pharmaceuticals and personal care products (PPCPs). Measurement of PPCPs has been of active interest in emerging and pre-regulatory practice for 20 years now, coinciding with the greater availability of LC-MS/MS systems in laboratories. The US EPA first showed interest in PPCPs in 2005 or thereabouts, leading us at SGS to develop and validate EPA 1694, a starter list of 73 PPCPs in aqueous and solid matrices and a companion EPA stability study. This method was then used to assess concentrations in biosolids from the EPA’s 2001 national sewage sludge survey.
Since then, we have expanded EPA 1694 to nearly 200 PPCPs and hormones and this approach has been used extensively in pre-regulatory testing by multiple regional and national agencies across North America. Measuring PPCPs is complicated, so we wanted to break this complexity down.
Measuring pharmaceuticals as a broad class

Drugs are not an analytical class of contaminants linked by common structural elements (PCBs, Dioxins, PFAS). So, they can vary from small molecules like acetaminophen (MW 151) to large peptide or protein drugs in the many thousands of Daltons. They can vary from relatively non-polar to very polar, relatively stable to very unstable. All in all, the search for a comprehensive single method for drugs in the environment is not going to be successful. Drugbank identifies nearly 20000 drugs, >15000 of them small-molecule though only a fraction are approved and in use. This, is, to use highly technical parlance, a lot. So, prioritization is key, as we will talk about.
The EPA 1694 approach for example focuses on small molecule organics that are amenable to capture on a hydrophilic-lipophilic balance (HLB) cartridge phase and measurement by LC-MS/MS. This covers a range of bases, acids and neutrals of varying polarity. However, it will not work on very small molecules, large molecules or very polar/non-polar ones. In developing EPA 1694, we used multiple extraction pHs and cartridge conditions to broaden chemical coverage, but there are limits.
PPCP Prioritization
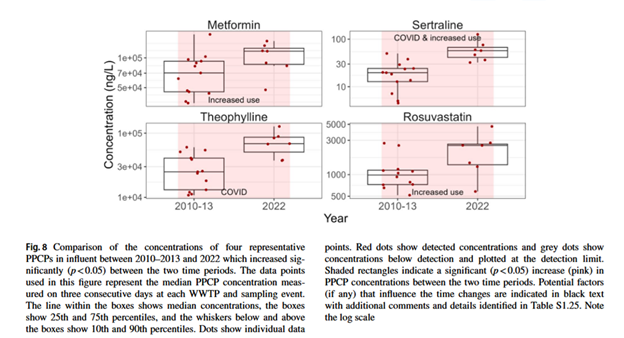
Figure 1: Image from Gewurtz et al. 2025 on the occurrence fate and time trends of PPCPs in Canadian Wastewater Systems
The drug universe is vast. So, how do we prioritize?
- The EPA has released some starting lists of pharmaceuticals to prioritize with interim health advisory levels provided. There are 697 targets (!) on this list with 374 of them listing Human Health Benchmarks (HHB-Rx). They range from sub ng/L (atropine) to many ug/L. At first glance our labs have experience with more than 90 of these PPCPs, and more if you look at metabolites etc. It’s a starting point, but quite an undertaking. Based on this long list, we probably need to prioritize even more.
- Based on occurrence? There is occurrence data for pharmaceuticals, especially the “HLB” ones. For example, Environment and Climate Change Canada (ECCC) has published extensively on the occurrence and fate of PPCPs through wastewater treatment using extended EPA 1694 approaches
- Based on toxicity and mode of action? For example, synthetic hormones can be biologically active at levels < 1 ng/L which are hard to measure. One of my favourite co-author publications was where we measured selective serotonin reuptake inhibitors (SSRIs) such as fluoxetine and sertraline, and neurotransmitter metabolites in fish exposed to wastewater effluent in a lagoon, and even at the low levels of exposure, the downstream fish were bolder
- Based on survival through the water treatment process? Pharmaceuticals unlike many PFAS undergo significant transformation and destruction through treatment. The ones that end up in finished drinking water will likely be the subset that is relatively stable/recalcitrant.
There are several research papers that go into the topic of prioritization using a combination of these and many other factors. Here’s one of them (Mo et al. 2022) that discusses various prioritization approaches.
Transformations and Stability
PPCPs are designed to be metabolized and excreted by the body. Frequently, measuring the parent drug alone is not sufficient. For example, you measure 2-hydroxy-ibuprofen along with ibuprofen. But it gets complicated. Theophylline is a bronchodilator drug, but also a caffeine metabolite. Hydrocortisone is used as a topical/oral drug, but you may know it as cortisol, that famous stress hormone! Many drugs also are excreted by the body by conjugation with glucuronide or sulfate to make them more polar. When we helped ECCC measure the influence of conjugation, they found that drug conjugates (especially hormones) can sometimes survive wastewater treatment and become an active source of the active drug in the environment.
Sample stability and the effect of chlorination is another major consideration. We did as part of our EPA support perform stability studies, assess the effects of free chlorine and use of dechlorinating agents. The EPA published this study in 2010 noting several insights and areas of remaining uncertainty and study. We did find that chlorination had the side effect of disappearing many drugs, though it wasn’t clear at the time whether this was destruction or transformation. There is a lot of uncertainty here! However, the next phase of PPCP monitoring must target metabolites and transformation products too.
The Role of Suspect Screening and Non-Target Analysis
The good news on drugs is that their structures, properties and behaviour are relatively well known known (we do have to study them extensively before approval) that suspect screening list approaches using high-resolution mass spectrometry that aim for broad coverage of the drugs and transformation products can, with some development and testing, perform the initial screening and prioritization. Increasingly, laboratories have added high-resolution LC-MS/MS capabilities. So, this approach becomes more feasible than even 10 years back. My belief is that LC-HRMS techniques can shine here (as long as we keep sample preparation in mind!). With the EPA providing a starting list, our task is potentially easier.

The Sample Preparation
My favorite warning to fellow measurement scientists is “think of the frontend too!”. Sophisticated instruments can only detect what’s in the final extract. The 1694 approach is ubiquitous and broad, but it’s not all encompassing. As we prioritize, we need to understand cases where a significant drug or transformation product may need alternate sample preparation/concentration processes. So, while HLB at multiple pHs is the go-to approach, we are actively working on expanding our chemical space through alternate chemistries.
Putting it all together
Existing fate and occurrence data combined with new studies, 1694-based methods and high-resolution suspect screening, combined with national drug use inventories could help us narrow our focus from the thousands into the 100s, picking the drugs and metabolites and transformation products that are
- Most likely to occur based on usage statistics
- Stable enough to survive treatment
- Of significant concern due to their activity, toxicity and mode of action
- Have history of occurrence in wastewater treatment, likely the main source of drugs into the environment through effluent and land-applied biosolids
In the interim, the EPA 1694 approach with its broad sweep is well-validated and easily implemented as a first pass for occurrence. However, there are significant uncertainties on stability, occurrence and other aspects of measurement that we need to deal with. We are really looking forward to helping the environmental community of practice on this issue.
Treatment is the big and expensive unknown, we’re not quite ready for this discussion yet even if there’s research level data on the fate of many pharmaceuticals through both basic secondary, and advanced treatment.
References
- U.S. Environmental Protection Agency. Draft contaminant candidate list 6 (CCL 6) [Internet]. Washington (DC): US EPA; 2026 Apr 2 [cited 2026 Apr 6]. Available from: https://www.epa.gov/ccl/draft-contaminant-candidate-list-6-ccl-6
- U.S. Environmental Protection Agency. Draft CCL 6 chemical contaminants [Internet]. Washington (DC): US EPA; 2026 [cited 2026 Apr 6]. Available from: https://www.epa.gov/ccl/draft-ccl-6-chemical-contaminants
- U.S. House of Representatives, Office of the Law Revision Counsel. 21 U.S.C. § 321: definitions; “drug” [Internet]. Washington (DC): US Government; [cited 2026 Apr 6]. Available from: https://uscode.house.gov/view.xhtml?req=(title:21%20section:321)
- SGS. Microplastics (emerging contaminants services): SGS; [cited 2026 Apr 6]. Available from: https://env.sgs.com/services/emerging-contaminants/microplastics/
- SGS. Pharmaceuticals and personal care products (PPCPs): SGS; [cited 2026 Apr 6]. Available from: https://env.sgs.com/services/emerging-contaminants/ppcps/
- U.S. Environmental Protection Agency. Method 1694 (pharmaceuticals and personal care products) frequent questions [Internet]. Washington (DC): US EPA; [cited 2026 Apr 6]. Available from: https://www.epa.gov/cwa-methods/method-1694-frequent-questions
- U.S. Environmental Protection Agency. Pharmaceuticals and personal care products (PPCPs) holding time study [Internet]. Washington (DC): US EPA; 2010 [cited 2026 Apr 6]. Available from: https://www.epa.gov/sites/default/files/2015-09/documents/ppcp-holding-time-study_2010.pdf
- McClellan K, Halden RU. Pharmaceuticals and personal care products in archived U.S. biosolids from the 2001 EPA national sewage sludge survey. Water Res. 2010;44(2):658-668. doi:10.1016/j.watres.2009.09.032
- DrugBank Online. Drug statistics [Internet]. Edmonton (AB): DrugBank; [cited 2026 Apr 6]. Available from: https://go.drugbank.com/stats
- Gewurtz SB, Auyeung AS, Teslic S, Smyth SA. Pharmaceuticals and personal care products in Canadian municipal wastewater and biosolids: occurrence, fate, and time trends 2010–2013 to 2022. Environ Sci Pollut Res Int. 2025;32:5022-5039. doi:10.1007/s11356-025-36007-0
- Simmons DBD, McCallum ES, Balshine S, Chandramouli B, Cosgrove J, Sherry JP. Reduced anxiety is associated with the accumulation of six serotonin reuptake inhibitors in wastewater treatment effluent exposed goldfish Carassius auratus. Sci Rep. 2017;7:17001. doi:10.1038/s41598-017-15989-z. PMID:29208964
- Mo, J., Guo, J., Iwata, H., Diamond, J., Qu, C., Xiong, J., & Han, J. (2024). What Approaches Should be Used to Prioritize Pharmaceuticals and Personal Care Products for Research on Environmental and Human Health Exposure and Effects? Environmental Toxicology and Chemistry, 43(3), 488–501. https://doi.org/10.1002/etc.5520
- Gewurtz SB, Teslic S, Hamilton MC, Smyth SA. Influence of conjugation on the fate of pharmaceuticals and hormones in Canadian wastewater treatment plants. ACS ES&T Water. 2022;2:329-338. doi:10.1021/acsestwater.1c00376

